2023, 8(3): 820-830.
doi: 10.1016/j.gee.2021.10.003
Abstract:
Various strategies, including controls of morphology, oxidation state, defect, and doping, have been developed to improve the performance of Cu-based catalysts for CO2 reduction reaction (CO2RR), generating a large amount of data. However, a unified understanding of underlying mechanism for further optimization is still lacking. In this work, combining first-principles calculations and machine learning (ML) techniques, we elucidate critical factors influencing the catalytic properties, taking Cu-based single atom alloys (SAAs) as examples. Our method relies on high-throughput calculations of 2669 CO adsorption configurations on 43 types of Cu-based SAAs with various surfaces. Extensive ML analyses reveal that low generalized coordination numbers and valence electron number are key features to determine catalytic performance. Applying our ML model with cross-group learning scheme, we demonstrate the model generalizes well between Cu-based SAAs with different alloying elements. Further, electronic structure calculations suggest surface negative center could enhance CO adsorption by back donating electrons to antibonding orbitals of CO. Finally, several SAAs, including PCu, AgCu, GaCu, ZnCu, SnCu, GeCu, InCu, and SiCu, are identified as promising CO2RR catalysts. Our work provides a paradigm for the rational design and fast screening of SAAs for various electrocatalytic reactions.





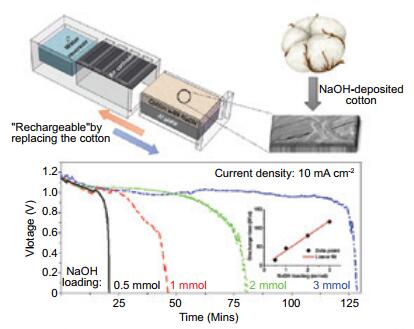
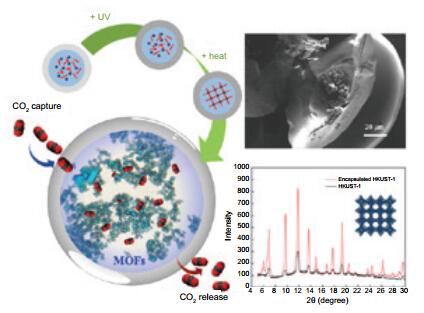
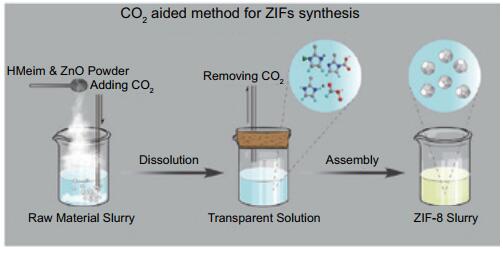
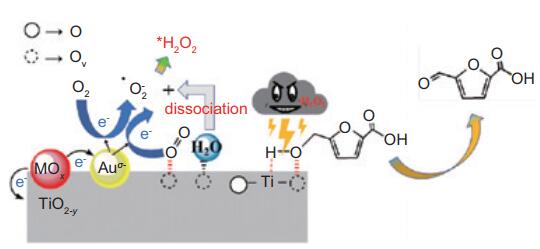





 京公网安备 11010802039050号
京公网安备 11010802039050号